Monday, 6:00 am
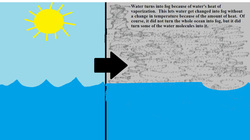
On Monday morning when we woke up, ready to begin our first day as a marine biologist, we were disappointed to see that our activities would have to be postponed until Tuesday. Everywhere was covered in fog! Unaware of why this occurred, we researched it and discovered that it was caused by heat of vaporization. Heat of vaporization is how much heat is required to change a liquid into a vapor without a temperature change. Fog is an example of this because it is formed from some of the ocean water molecules turning into fog, due to heat of vaporization.
On Monday morning when we woke up, ready to begin our first day as a marine biologist, we were disappointed to see that our activities would have to be postponed until Tuesday. Everywhere was covered in fog! Unaware of why this occurred, we researched it and discovered that it was caused by heat of vaporization. Heat of vaporization is how much heat is required to change a liquid into a vapor without a temperature change. Fog is an example of this because it is formed from some of the ocean water molecules turning into fog, due to heat of vaporization.

Tuesday, 6:00 am
When we woke up Tuesday morning, we went onto the balcony to enjoy the sunrise. As we stepped outside, we realized that it was raining. Staring at the raindrops as they fell over the ocean, the water in the raindrops stayed together. This is because of cohesion. Cohesion is water sticking to water, because water is attracted to itself and will come together whenever possible. It amazed us that the raindrops stuck together instead of just splatting to the ground. We could make out individual raindrops, and knew that within every drop there was water sticking together, thanks to cohesion.
When we woke up Tuesday morning, we went onto the balcony to enjoy the sunrise. As we stepped outside, we realized that it was raining. Staring at the raindrops as they fell over the ocean, the water in the raindrops stayed together. This is because of cohesion. Cohesion is water sticking to water, because water is attracted to itself and will come together whenever possible. It amazed us that the raindrops stuck together instead of just splatting to the ground. We could make out individual raindrops, and knew that within every drop there was water sticking together, thanks to cohesion.
Tuesday, 12:00 pm
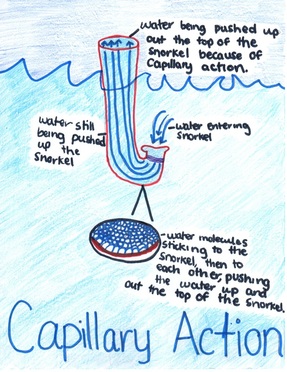
Today we had our first snorkeling experience! Since we had no idea how to do it, we did it wrong and let go of the mouthpiece, so the water got into the tube. It surprised us because even though we weren’t very far under the water, the water came out the top of the snorkel. We finally realized after a couple of times of trying it again, that capillary action makes it do that. The water sticks to the side of the snorkel, and more water sticks to itself, until it climbs its way out of the snorkel. This was pretty cool, because we never knew that. As it turns out, it’s also the reason that the plants get their water; by capillary action!
Today we had our first snorkeling experience! Since we had no idea how to do it, we did it wrong and let go of the mouthpiece, so the water got into the tube. It surprised us because even though we weren’t very far under the water, the water came out the top of the snorkel. We finally realized after a couple of times of trying it again, that capillary action makes it do that. The water sticks to the side of the snorkel, and more water sticks to itself, until it climbs its way out of the snorkel. This was pretty cool, because we never knew that. As it turns out, it’s also the reason that the plants get their water; by capillary action!

Tuesday, 2:00 pm
After a quick lunch at the Sea Shack we headed back down to the shore to take a closer look at the seals. We got there at the perfect time; they were just coming out of the water. As they propped themselves up on the rocks we noticed something strange, the water was sticking to them. Curious as to why they were still wet, we further investigated and came to two conclusions. The water was sticking to the seals because of adhesion. Adhesion is when water sticks to other substances because it is drawn to other polar substances. This leads up to the other reason that the water was sticking to the seal, polarity. Polarity is unequal sharing of electrons within a molecule that causes strong attraction to similar substances. The seal is polar, so water sticks to the seal because of adhesion and polarity.
After a quick lunch at the Sea Shack we headed back down to the shore to take a closer look at the seals. We got there at the perfect time; they were just coming out of the water. As they propped themselves up on the rocks we noticed something strange, the water was sticking to them. Curious as to why they were still wet, we further investigated and came to two conclusions. The water was sticking to the seals because of adhesion. Adhesion is when water sticks to other substances because it is drawn to other polar substances. This leads up to the other reason that the water was sticking to the seal, polarity. Polarity is unequal sharing of electrons within a molecule that causes strong attraction to similar substances. The seal is polar, so water sticks to the seal because of adhesion and polarity.

Tuesday, 4:00 pm
This afternoon, we went down to the beach and we saw a little fish slide under a rock in a tidal pool. We lifted up the rock, and so that none of the other animals got hurt, we tossed the rock into the ocean. Surprisingly enough, it skipped over the water a few times, then it finally sunk. After puzzling over this trivial fact for a minute, we learned that it was because of surface tension. Surface tension is when it seems like there is something on the surface of the water, holding the water molecules together. When 2 hydrogen atoms and one oxygen atom are combined, it creates a water molecule. If there is a lot of water, it takes a lot more force for something to break the surface tension. Surface tension made the rock not sink as fast as we assumed it would, because the molecules were holding together strong, and it took the force of the rock to break the surface. It’s amazing what we have already learned on the trip so far, and it’s only the first day!
This afternoon, we went down to the beach and we saw a little fish slide under a rock in a tidal pool. We lifted up the rock, and so that none of the other animals got hurt, we tossed the rock into the ocean. Surprisingly enough, it skipped over the water a few times, then it finally sunk. After puzzling over this trivial fact for a minute, we learned that it was because of surface tension. Surface tension is when it seems like there is something on the surface of the water, holding the water molecules together. When 2 hydrogen atoms and one oxygen atom are combined, it creates a water molecule. If there is a lot of water, it takes a lot more force for something to break the surface tension. Surface tension made the rock not sink as fast as we assumed it would, because the molecules were holding together strong, and it took the force of the rock to break the surface. It’s amazing what we have already learned on the trip so far, and it’s only the first day!

Tuesday, 7:30 pm
Today, while walking on the beach, we saw a shell that had been worn down by the ocean. We wondered how it had been broken down and partially dissolved, and it turns out, the answer is because water is a universal solvent. Water is the universal solvent in the fact that it breaks down and dissolves more things than most liquids can, but nothing can dissolve everything. After years, it finally wore down the shell and dissolved bits and parts, but it is a very long process for something so big, which is why the shell was not completely dissolved.
Today, while walking on the beach, we saw a shell that had been worn down by the ocean. We wondered how it had been broken down and partially dissolved, and it turns out, the answer is because water is a universal solvent. Water is the universal solvent in the fact that it breaks down and dissolves more things than most liquids can, but nothing can dissolve everything. After years, it finally wore down the shell and dissolved bits and parts, but it is a very long process for something so big, which is why the shell was not completely dissolved.

Wednesday, 11:00 am
We went for a quick swim around 11 to explore the different marine life. Wading into the tide, a question occurred to me. How do we swim in the ocean? What causes the water to move? I asked this to my partner and we discussed the different aspects of hydrogen bonds. A hydrogen bond is an easily formed bond. The polarity of water causes it to bond, but the hydrogen bonds are very weak and have little energy. When we swam in the ocean, we broke the hydrogen bonds by simply stepping in, walking around, and swimming. The simple touch of a finger can break a hydrogen bond, because while they are very important in keeping water together, they are very able to break.
We went for a quick swim around 11 to explore the different marine life. Wading into the tide, a question occurred to me. How do we swim in the ocean? What causes the water to move? I asked this to my partner and we discussed the different aspects of hydrogen bonds. A hydrogen bond is an easily formed bond. The polarity of water causes it to bond, but the hydrogen bonds are very weak and have little energy. When we swam in the ocean, we broke the hydrogen bonds by simply stepping in, walking around, and swimming. The simple touch of a finger can break a hydrogen bond, because while they are very important in keeping water together, they are very able to break.
Thursday, 9:00 am
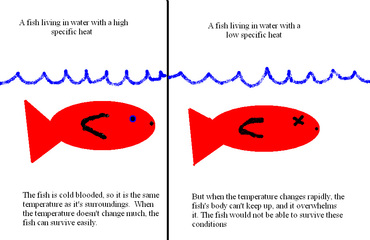
Today we decided to go out and look at the fish and other water species early in the morning. It was beautiful, watching the sun rise over the ocean, while we waded into it for some early morning swimming. Oddly enough, the water wasn’t much cooler than it was yesterday, even though the temperature dropped 9 degrees over the course of the night. This is because of specific heat. The water absorbs the heat from the sun during the day, and then slowly releases it, so that the temperature doesn’t change much. It may not seem like a big deal, but it is. Fish and some other water species are cold blooded, so they become the same temperature
as their surroundings. A high drop or rise in temperature in a short period of time would be the death of all of these animals, so it’s good that the specific heat for water is so high.
Today we decided to go out and look at the fish and other water species early in the morning. It was beautiful, watching the sun rise over the ocean, while we waded into it for some early morning swimming. Oddly enough, the water wasn’t much cooler than it was yesterday, even though the temperature dropped 9 degrees over the course of the night. This is because of specific heat. The water absorbs the heat from the sun during the day, and then slowly releases it, so that the temperature doesn’t change much. It may not seem like a big deal, but it is. Fish and some other water species are cold blooded, so they become the same temperature
as their surroundings. A high drop or rise in temperature in a short period of time would be the death of all of these animals, so it’s good that the specific heat for water is so high.

Thursday, 8:15 am
Today we were out looking at the penguins! They live in very cold regions, and there is a ton of ice in their habitat. As we were out looking at them, we realized this: The ice didn’t sink to the bottom, yet if we were to drop something in that weighed as much as the iceberg did, it definitely would. After explaining my ideas to my partner, she agreed that it was definitely peculiar. We finally figured it out, and it turns out the it is because of density! Density and the specific heat of the water are vital to letting it float. The density of ice is actually less than that of regular water, by .08 grams per milliliter. You see, the water molecules spread out more in ice than they do in water, and since it has less space, it is less dense. The ice then floats on the top. Penguins are definitely fascinating, but water is definitely getting more and more interesting the more we learn about it!
